Data governance and informed consent
02 November 2017
The responsible use of data will be discussed at an HBP organised seminar next month in Paris.
In an age of big data, questions about consent and data protection are extremely important, says Lise Bitsch, manager of the Human Brain Project’s Ethics and Society Subproject, which is organising the event.
“We believe a solid ethical framework for the type of big data research developed by the HBP requires a dialogue involving a wide variety of stakeholders such as patient representatives, personal data managers, bioethicists and healthcare providers. The present workshop is therefore organised with the purpose of bringing the voices of these groups into the HBP development process.”
These issues are particularly relevant to data on patients and research participants that are stored in hospitals around the world. The data could be brain images, previously received therapies, mental evaluation, or genetic information. Such data provides a valuable resource for research, if it can be accessed responsibly.
The seminar will bring together leading neurologists, personal data managers, experts in bioethics and patient representatives to discuss the challenges setting up and participating in ‘big data’ research.
The aim of the seminar is to initiate a dialogue between the Human Brain Project (HBP) scientists, HBP data managers and external experts and stakeholders, on how to improve data governance and informed consent. The outcome of the seminar will include recommendations for the research community and concrete input to the HBP Standard Operating Procedures (SOPs) and data policy manuals.
In particular, the seminar will focus on the following questions:
- Which options exists for obtaining informed consent in big data research?
- What can we learn from biobanks and brain image databases about data governance?
- How do we empower of data subjects in ‘big data research’?
- Which options lies in privacy by design?
- How can informed consent procedures be improved for unconscious people?
- How do we balance data protection and informed consent against other societal values?
- How can we develop standard procedures for collecting and researching on clinical data in big data projects?
The seminar will take place on 11-12 December at the European Institute for Theoretical Neuroscience (EITN), 74 Rue du Faubourg Saint-Antoine, 75012 Paris, France. The deadline for registration is 11 November.
More information can be found by downloading the
![]() invite and programme. (972.0 KB)
invite and programme. (972.0 KB)
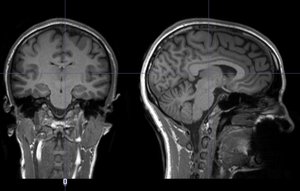
An MRI image of a healthy brain. Image courtesy: Centre Hospitalier Universitaire Vaudois.



